Comportement, plasticité et mémoire du poisson zèbre
Chef d’équipe : Owen RANDLETT — Team website
Neurobiologie | Poisson zèbre | Mémoire | Habituation | O-Bend | Nf1
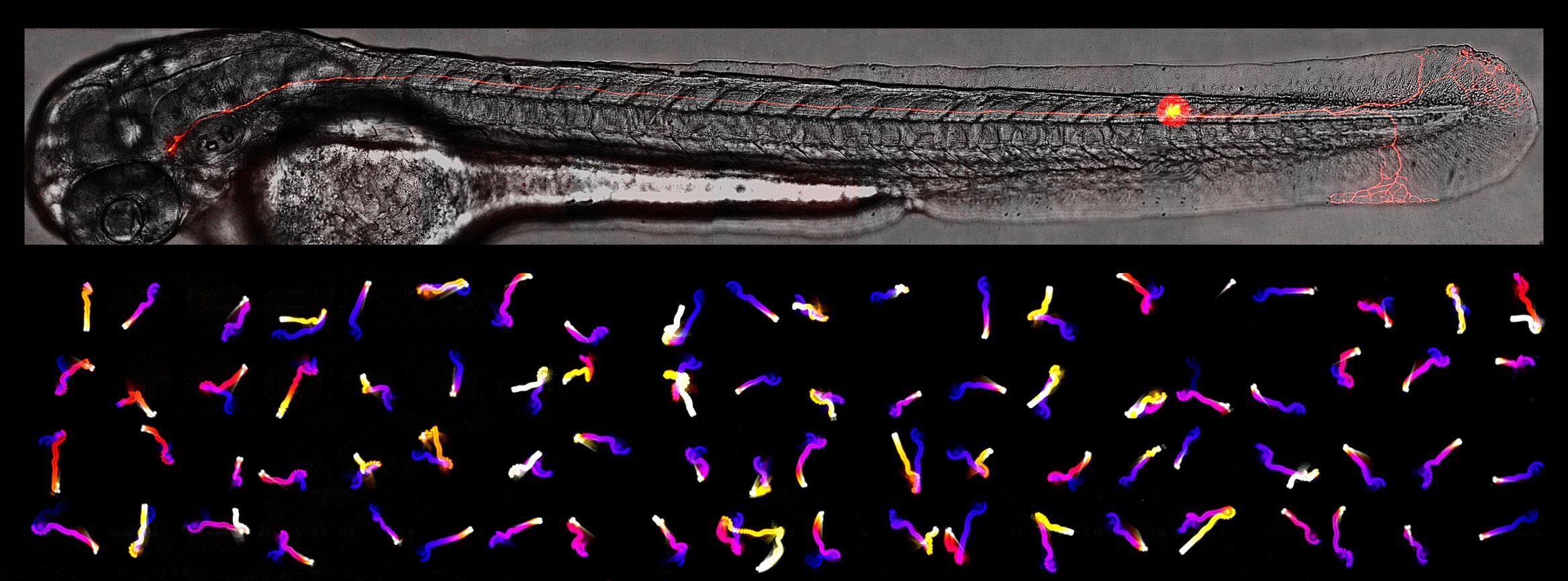
 We are interested in how the brain generates simple behaviours, and how its changes with experience to alter behaviour.
We are interested in how the brain generates simple behaviours, and how its changes with experience to alter behaviour.
We work with larval zebrafish, which are a very small and transparent model vertebrate. Despite being less than a week old, zebrafish exhibit sophisticated behaviours, which we are trying to understand using a combination of genetics and functional neuroscience approaches.
Our work focuses on three related questions:
1. The mechanisms of plasticity that lead to long-term memory.
We use paradigms to train larvae to ignore repeated stimuli. This simple form of learning is known as habituation, and offers a tractable paradigm to study the general phenomenon of learning and memory. Despite the apparent triviality of habituation (simply learning to ignore a given stimulus), how the brain actually accomplishes this selective filtration of specific stimuli is still largely unclear. Indeed, we have shown that habituation is a complex phenomenon that involves multiple independent plasticity events that each tune individual components of behaviour. We hope to gain insights into this process at the molecular, cellular and circuit levels.
2. Visual navigation
When presented with a choice, zebrafish larvae will swim towards lit environments and avoid darkness. We are interested in understanding the aspects of such stimuli that are attractive/repulsive to the larvae, how this manifests in brain activity patterns, and how this directs swimming behaviour resulting in directed navigation.
3. Zebrafish models of neurological disorders
As a vertebrate the genetics and neuroanatomy of zebrafish show many homologies to humans. Therefore, it may be possible to model human genetic disorders in zebrafish in order to study the basic biology of affected genetic/molecular pathways, and high-throughput approaches to identify novel therapeutics. We are currently focusing on Autosomal Recessive Cerebellar Ataxias, to determine if/how genes linked to these disorders in humans manifest in behavioural and cerebellar-dependent phenotypes in larval zebrafish, and what this might tell us about the biology underlying ataxia.
Membres de l’équipe
- Owen RANDLETT — CR, INSERM, HDR
owen.randlett@univ-lyon1.fr – 04 78 77 70 16 - Dominique BAAS — MCU, UCBL
dominique.baas@univ-lyon1.fr – 04 78 77 28 71 - Abdel RAHMAN EL HASSAN — Doctorant, CNRS
abdel-rahman.el-hassan@univ-lyon1.fr – 04 78 77 28 71 - Adrià MARTÍNEZ PÉREZ — Doctorant, UCBL
– 04 78 77 28 71 - Julien PERRICHET — AI, UCBL – julien.perrichet@univ-lyon1.fr – 04 78 77 28 65
Sélection de publications
Estradiol Promotes Habituation Learning via an Unidentified Target, Bypassing the Suppressive Effects of Established ERs
Hsiao A., Darvaux-Hubert I., Hicks D., et al.. 🔗 https://doi.org/10.1210/endocr/bqaf110
Résumé :
Abstract Habituating to the constant stimuli in the environment is a critical learning process conserved across species. We use a larval zebrafish visual response to sudden darkness as a model for studying habituation learning, where zebrafish reduce their responses to repeated stimulations. In this paradigm, treatment with estradiol strongly increases learning rate, resulting in more strongly suppressed responses. We used mutant lines for the estrogen receptors (ERs)—esr1, esr2a, esr2b, gper1—in an attempt to identify the receptor(s) mediating these effects. These experiments failed to identify a necessary receptor (or combination of receptors). Surprisingly, esr1, esr2a, and gper1 mutants showed weak but consistent increases in habituation, indicating that these receptors suppress habituation learning. These experiments demonstrate that estradiol is a complex modulator of learning in our model, where the learning-promoting effects are mediated by an unidentified estradiol target, and the classical estrogen receptors act in competition to subtly suppress learning.
Endocrinology 166, (2025)
pi_tailtrack: A compact, inexpensive and open-source behaviour-tracking system for head-restrained zebrafish
Randlett O.. 🔗 https://doi.org/10.1242/jeb.246335
Résumé :
ABSTRACT Quantifying animal behaviour during microscopy is crucial to associate optically recorded neural activity with behavioural outputs and states. Here, I describe an imaging and tracking system for head-restrained larval zebrafish compatible with functional microscopy. This system is based on the Raspberry Pi computer, Pi NoIR camera and open-source software for the real-time tail segmentation and skeletonization of the zebrafish tail at over 100 Hz. This allows for precise and long-term analyses of swimming behaviour, which can be related to functional signals recorded in individual neurons. This system offers a simple but performant solution for quantifying the behaviour of head-restrained larval zebrafish, which can be built for 340€.
Journal of Experimental Biology 226, (2023)
Functional and pharmacological analyses of visual habituation learning in larval zebrafish
Lamiré L., Haesemeyer M., Engert F., et al.. 🔗 https://doi.org/10.7554/elife.84926.3
Résumé :
Habituation allows animals to learn to ignore persistent but inconsequential stimuli. Despite being the most basic form of learning, a consensus model on the underlying mechanisms has yet to emerge. To probe relevant mechanisms, we took advantage of a visual habituation paradigm in larval zebrafish, where larvae reduce their reactions to abrupt global dimming (a dark flash). We used Ca 2+ imaging during repeated dark flashes and identified 12 functional classes of neurons that differ based on their rate of adaptation, stimulus response shape, and anatomical location. While most classes of neurons depressed their responses to repeated stimuli, we identified populations that did not adapt or that potentiated their response. These neurons were distributed across brain areas, consistent with a distributed learning process. Using a small-molecule screening approach, we confirmed that habituation manifests from multiple distinct molecular mechanisms, and we have implicated molecular pathways in habituation, including melatonin, oestrogen, and GABA signalling. However, by combining anatomical analyses and pharmacological manipulations with Ca 2+ imaging, we failed to identify a simple relationship between pharmacology, altered activity patterns, and habituation behaviour. Collectively, our work indicates that habituation occurs via a complex and distributed plasticity processes that cannot be captured by a simple model. Therefore, untangling the mechanisms of habituation will likely require dedicated approaches aimed at sub-component mechanisms underlying this multidimensional learning process.
eLife 12, (2023)
Financements et soutien












