NEST – Embryogénèse du Système Nerveux et étude des Tumeurs pédiatriques
Chef d’équipe : Valérie CASTELLANI
Développement embryonnaire | cancers pédiatriques | neuroblastome | médulloblastome | métastases | migration cellulaire | microenvironnement | guidage axonal | crêtes neurales | système nerveux entérique | cortex | moelle épinière | modèle aviaire | transcriptomique en cellule unique | microscopie à feuillet de lumière
 Nous étudions les mécanismes qui sous-tendent le neuro-développement embryonnaire et les tumeurs pédiatriques malignes d’origine prénatale.
Nous étudions les mécanismes qui sous-tendent le neuro-développement embryonnaire et les tumeurs pédiatriques malignes d’origine prénatale.
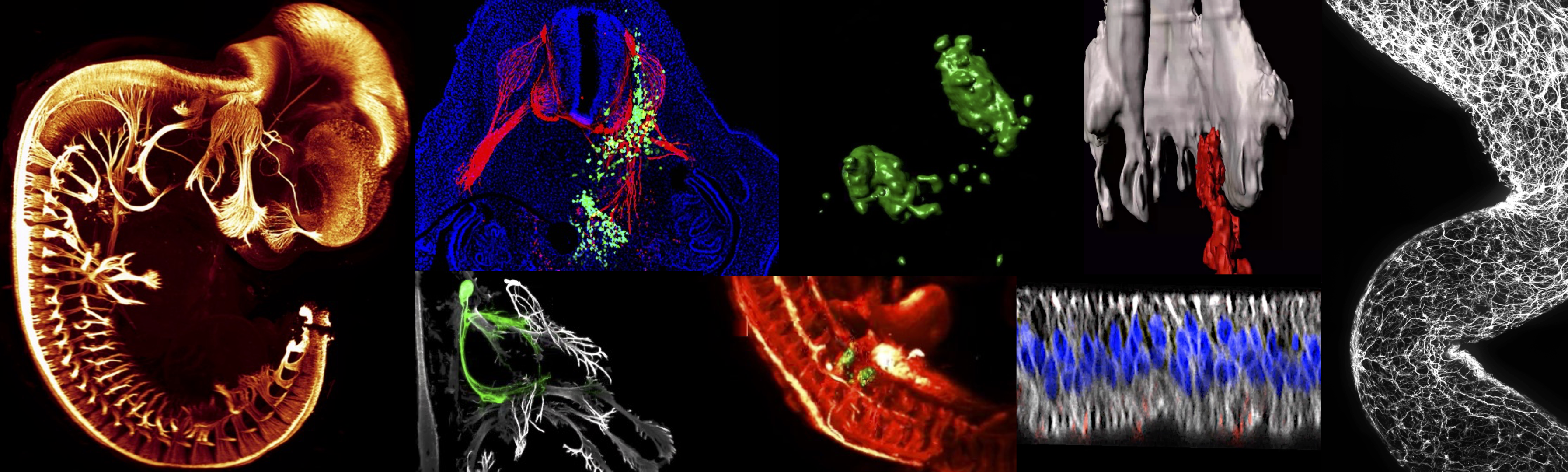
Notre laboratoire étudie les mécanismes cellulaires et moléculaires qui contrôlent la formation du système nerveux chez l’embryon. Nous nous intéressons à la communication des cellules progénitrices et neuronales avec leur environnement, et plus particulièrement aux processus cellulaires et signalisations moléculaires qui dirigent la colonisation des territoires embryonnaires par les cellules neurales et les axones en migration. Nous abordons actuellement ces questions en étudiant une population remarquable de cellules embryonnaires, la crête neurale, à l’origine de multiples dérivés, dont la chaîne sympathique, la glande médullosurrénale et le système nerveux entérique. En combinant des manipulations expérimentales dans le modèle de l’embryon aviaire et les analyses transcriptomiques en cellule unique, nous étudions l’émergence des circuits neuronaux entériques et la connectivité cerveau-intestin pour caractériser la dynamique d’émergence de la diversité neuronale et les programmes génétiques qui médient les dialogues entre les cellules et leur environnement. Nous cherchons également à savoir si ces programmes sont conservés dans l’embryon humain et si leur dérégulation pourrait contribuer à certaines pathologies neurodéveloppementales.
En parallèle, nous étudions les cancers pédiatriques à la lumière de leur origine embryonnaire, en particulier le neuroblastome dérivé de la crête neurale et le médulloblastome dérivé des précurseurs cérébelleux. L’hétérogénéité et la plasticité qui caractérisent ces tumeurs malignes et sous-tendent leur agressivité seraient enracinées dans le contexte embryonnaire de leur émergence. Les événements tumorigènes se produisent dans des cellules qui communiquent activement avec leur environnement et qui sont dotées des propriétés prolifératives et migratoires nécessaires à la formation des tissus. Tout en devenant malignes, les cellules tumorales conservent de nombreuses caractéristiques des cellules d’origine. Notre objectif est de comprendre comment ce double potentiel physiologique et tumorigène se manifeste au cours de la tumorigenèse et la progression métastatique par l’exploitation opportuniste ou le détournement des mécanismes du développement et de ses signalisations moléculaires. Pour répondre à ces questions, et en tirant parti de nos modèles de biologie du développement, nous avons établi un paradigme in vivo spécifique qui récapitule le contexte embryonnaire d’émergence de ces cancers. Il consiste à transplanter des cellules tumorales humaines dans des tissus sélectionnés de l’embryon aviaire. Nos études sont basées sur une stratégie multi-approche combinant l’embryologie expérimentale, les études fonctionnelles des gènes d’intérêt dans les modèles aviaires, la microscopie à nappe de lumière 3D pour cartographier les cellules et les molécules au niveau de l’embryon entier, la vidéomicroscopie. Cette recherche fondamentale ouvrira la voie au développement de thérapies ciblant spécifiquement le comportement et la signalisation propres aux cellules malignes
Pour le grand Public
Comment se construisent nos circuits neuronaux ? Le neurone nouveau-né développe un prolongement, l’axone, destiné à entreprendre un incroyable voyage, à la recherche des cellules avec lesquelles il établira une communication. Ainsi, au cours du développement embryonnaire et post-natal, des millions d’axones partent à la recherche de leurs partenaires, certains restant confinés dans le cerveau ou la moelle épinière, d’autres colonisant l’ensemble de l’organisme pour innerver les muscles, la peau, les viscères. De nombreuses cellules neurales effectuent également des migrations pour coloniser des territoires distaux dans lesquels elles construiront une structure nerveuse. Des signaux moléculaires sont exprimés dans les tissus embryonnaires qui permettent aux axones et aux cellules de se localiser dans l’espace. Ils sont appelés signaux topographiques ou signaux de guidage. Amener chaque axone et chaque cellule à sa destination est un véritable défi et ce sont les mécanismes qui contrôlent ce processus que notre équipe étudie principalement. Les axones et les cellules peuvent-ils se perdre en chemin ou se tromper de destination ? Diverses pathologies infantiles résultent d’altérations dans la navigation des cellules et des axones. Il en existe sans doute beaucoup plus, mais identifier des altérations de ces mécanismes précoces reste encore difficile. Les cellules peuvent également devenir malignes à un stade prénatal, alors qu’elles sont en phase de migration et de prolifération. Elles donnent naissance à des tumeurs qui peuvent être très disséminées et dont l’évolution est sombre. Notre équipe étudie le comportement des cellules devenues malignes et la façon dont elles communiquent avec les tissus embryonnaires pendant la formation de la tumeur et la progression métastatique.
South-ROCK

Notre équipe est membre du centre d’excellence de recherche intégré South-ROCK en oncologie pédiatrique de Lyon-Marseille, l’un des trois lauréats de l’appel à projets PEDIACRIEX de l’Institut national français du cancer.
REACT4KIDS

Notre équipe fait partie de REACT4KIDS (REsearchers in oncology ACTing for kids), un réseau national de laboratoires de recherche en oncologie pédiatrique dont l’objectif est de favoriser les travaux collaboratifs pour accélérer les connaissances sur la biologie des cancers de l’enfant afin d’ouvrir la voie à des thérapies innovantes adaptées à ces cancers.
Start Up

Oncofactory est une spin-off du laboratoire de V. Castellani co-fondée par C. Delloye-Bourgeois et V. Castellani, qui exploite une plateforme technologique innovante permettant de créer des répliques miniaturisées de cancers de patients à partir de biopsies et chirurgies, dans un organisme embryonnaire aviaire, d’administrer des thérapies, d’imager les réponses tumorales par microscopie 3D dans l’organisme entier, et de réaliser des analyses moléculaires à large échelle sur les répliques tumorales. Oncofactory utilise sa plateforme pour accompagner les industries pharmaceutiques dans leurs programmes précliniques en oncologie.
Membres de l’équipe

- Valérie CASTELLANI — DRCE CNRS, HDR – valerie.castellani@univ-lyon1.fr
- Frédéric MORET — MCU, UCBL, HDR – frederic.moret@univ-lyon1.fr
- Julien FALK — CRCN, CNRS, HDR – julien.falk@univ-lyon1.fr
- Servane TAUSZIG-DELAMASURE — DR2, CNRS, HDR – servane.tauszig-delamasure@univ-lyon1.fr
- Muriel BOZON — AI, CNRS – muriel.bozon@univ-lyon1.fr
- Karine THOINET — AI, CNRS – karine.thoinet@univ-lyon1.fr
- Franck BOISMOREAU — Post-doctorant, UCBL – franck.boismoreau@univ-lyon1.fr
- Audrey PRUNET — Post-doctorante – audrey.prunet@univ-lyon1.fr
- Claire RIVET — Doctorante – claire.rivet@univ-lyon1.fr
- Marion MALLET — Doctorante – marion.mallet@univ-lyon1.fr
- Luce ROSEIRO — Doctorante – luce.roseiro@univ-lyon1.fr
- Florian MARTIN — IE – florian.martin@univ-lyon1.fr
- Maëlys ANDRE — IE – maelys.andre@univ-lyon1.fr
- Julie PIGNOLET — IR — julie.pignolet@univ-lyon1.fr
- Baptiste Mida — IR — baptiste.mida@univ-lyon1.fr
- Myriam BOUDISSA — IE, myriam.boudissa@univ-lyon1.fr
- Cécile FAURE-CONTER — Oncopédiatre IHOPe, CLB – cecile.conter@ihope.fr
- Jérémy GANOFSKY — IE Bioinformaticien – jeremy.ganofsky@univ-lyon1.fr
Alumni
- Céline Delloye-Bourgeois — Chef d’équipe, Centre de recherche en Cancérologie de Lyon, France
- Homaira Nawabi — Chef d’équipe, Grenoble Institute of Neuroscience, France
- Camille Charoy — Senior microscopist at The Francis Crick Institute, UK
- Florie Reynaud — IR, Centre de recherche en Cancérologie de Lyon, France
- Aurora Pignata — MCU, Université de Toulouse, France
- Leila Boubakar — Post-doctorante, KU Leuven, Belgique
- Elise Arbeille — MCU, Université de Méditerranée, Marseille, France
- Anne Briançon-Marjollet — PU, Université Grenoble-Alpes , Grenoble, France
- Hugo Ducuing — Post-doctorant, Institut de Recherches Cliniques de Montréal, Canada
- Sarah Dinvaut —Faculté de médecine de Rockefeller, Lyon, France
Sélection de publications
Neuroblastoma plasticity during metastatic progression stems from the dynamics of an early sympathetic transcriptomic trajectory
Villalard B., Boltjes A., Reynaud F., et al.. 🔗 https://doi.org/10.1038/s41467-024-53776-3
Résumé :
Résumé non disponible.
Nature Communications 15, (2024)
GPC3-Unc5 receptor complex structure and role in cell migration
Akkermans O., Delloye-Bourgeois C., Peregrina C., et al.. 🔗 https://doi.org/10.1016/j.cell.2022.09.025
Résumé :
Résumé non disponible.
Cell 185, 3931-3949.e26 (2022)
Functional precision oncology for follicular lymphoma with patient-derived xenograft in avian embryos
Zala M., Lipinski B., Costechareyre C., et al.. 🔗 https://doi.org/10.1038/s41375-024-02150-9
Résumé :
Résumé non disponible.
Leukemia 38, 430-434 (2024)
Axon guidance during mouse central nervous system regeneration is required for specific brain innervation
Delpech C., Schaeffer J., Vilallongue N., et al.. 🔗 https://doi.org/10.1016/j.devcel.2024.09.005
Résumé :
Résumé non disponible.
Developmental Cell 59, 3213-3228.e8 (2024)
Motor innervation directs the correct development of the mouse sympathetic nervous system
Erickson A., Motta A., Kastriti M., et al.. 🔗 https://doi.org/10.1038/s41467-024-51290-0
Résumé :
AbstractThe sympathetic nervous system controls bodily functions including vascular tone, cardiac rhythm, and the “fight-or-flight response”. Sympathetic chain ganglia develop in parallel with preganglionic motor nerves extending from the neural tube, raising the question of whether axon targeting contributes to sympathetic chain formation. Using nerve-selective genetic ablations and lineage tracing in mouse, we reveal that motor nerve-associated Schwann cell precursors (SCPs) contribute sympathetic neurons and satellite glia after the initial seeding of sympathetic ganglia by neural crest. Motor nerve ablation causes mispositioning of SCP-derived sympathoblasts as well as sympathetic chain hypoplasia and fragmentation. Sympathetic neurons in motor-ablated embryos project precociously and abnormally towards dorsal root ganglia, eventually resulting in fusion of sympathetic and sensory ganglia. Cell interaction analysis identifies semaphorins as potential motor nerve-derived signaling molecules regulating sympathoblast positioning and outgrowth. Overall, central innervation functions both as infrastructure and regulatory niche to ensure the integrity of peripheral ganglia morphogenesis.
Nature Communications 15, (2024)
3D exploration of gene expression in chicken embryos through combined RNA fluorescence in situ hybridization, immunofluorescence, and clearing
André M., Dinvaut S., Castellani V., et al.. 🔗 https://doi.org/10.1186/s12915-024-01922-0
Résumé :
Abstract Background Fine characterization of gene expression patterns is crucial to understand many aspects of embryonic development. The chicken embryo is a well-established and valuable animal model for developmental biology. The period spanning from the third to sixth embryonic days (E3 to E6) is critical for many organ developments. Hybridization chain reaction RNA fluorescent in situ hybridization (HCR RNA-FISH) enables multiplex RNA detection in thick samples including embryos of various animal models. However, its use is limited by tissue opacity. Results We optimized HCR RNA-FISH protocol to efficiently label RNAs in whole mount chicken embryos from E3.5 to E5.5 and adapted it to ethyl cinnamate (ECi) tissue clearing. We show that light sheet imaging of HCR RNA-FISH after ECi clearing allows RNA expression analysis within embryonic tissues with good sensitivity and spatial resolution. Finally, whole mount immunofluorescence can be performed after HCR RNA-FISH enabling as exemplified to assay complex spatial relationships between axons and their environment or to monitor GFP electroporated neurons. Conclusions We could extend the use of HCR RNA-FISH to older chick embryos by optimizing HCR RNA-FISH and combining it with tissue clearing and 3D imaging. The integration of immunostaining makes possible to combine gene expression with classical cell markers, to correlate expressions with morphological differentiation and to depict gene expressions in gain or loss of function contexts. Altogether, this combined procedure further extends the potential of HCR RNA-FISH technique for chicken embryology.
BMC Biology 22, (2024)
An in vivo avian model of human melanoma to perform rapid and robust preclinical studies
Jarrosson L., Dalle S., Costechareyre C., et al.. 🔗 https://doi.org/10.15252/emmm.202216629
Résumé :
AbstractMetastatic melanoma patients carrying a BRAFV600 mutation can be treated with a combination of BRAF and MEK inhibitors (BRAFi/MEKi), but innate and acquired resistance invariably occurs. Predicting patient response to targeted therapies is crucial to guide clinical decision. We describe here the development of a highly efficient patient‐derived xenograft model adapted to patient melanoma biopsies, using the avian embryo as a host (AVI‐PDXTM). In this in vivo paradigm, we depict a fast and reproducible tumor engraftment of patient samples within the embryonic skin, preserving key molecular and phenotypic features. We show that sensitivity and resistance to BRAFi/MEKi can be reliably modeled in these AVI‐PDXTM, as well as synergies with other drugs. We further provide proof‐of‐concept that the AVI‐PDXTM models the diversity of responses of melanoma patients to BRAFi/MEKi, within days, hence positioning it as a valuable tool for the design of personalized medicine assays and for the evaluation of novel combination strategies.
EMBO Molecular Medicine 15, (2023)
The Neurod1/4-Ntrk3-Src pathway regulates gonadotrope cell adhesion and motility
Le Ciclé C., Pacini V., Rama N., et al.. 🔗 https://doi.org/10.1038/s41420-023-01615-7
Résumé :
AbstractPituitary gonadotrope cells are essential for the endocrine regulation of reproduction in vertebrates. These cells emerge early during embryogenesis, colonize the pituitary glands and organize in tridimensional networks, which are believed to be crucial to ensure proper regulation of fertility. However, the molecular mechanisms regulating the organization of gonadotrope cell population during embryogenesis remain poorly understood. In this work, we characterized the target genes of NEUROD1 and NEUROD4 transcription factors in the immature gonadotrope αT3-1 cell model by in silico functional genomic analyses. We demonstrated that NEUROD1/4 regulate genes belonging to the focal adhesion pathway. Using CRISPR/Cas9 knock-out approaches, we established a double NEUROD1/4 knock-out αT3-1 cell model and demonstrated that NEUROD1/4 regulate cell adhesion and cell motility. We then characterized, by immuno-fluorescence, focal adhesion number and signaling in the context of NEUROD1/4 insufficiency. We demonstrated that NEUROD1/4 knock-out leads to an increase in the number of focal adhesions associated with signaling abnormalities implicating the c-Src kinase. We further showed that the neurotrophin tyrosine kinase receptor 3 NTRK3, a target of NEUROD1/4, interacts physically with c-Src. Furthermore, using motility rescue experiments and time-lapse video microscopy, we demonstrated that NTRK3 is a major regulator of gonadotrope cell motility. Finally, using a Ntrk3 knock-out mouse model, we showed that NTRK3 regulates gonadotrope cells positioning in the developing pituitary, in vivo. Altogether our study demonstrates that the Neurod1/4-Ntrk3-cSrc pathway is a major actor of gonadotrope cell mobility, and thus provides new insights in the regulation of gonadotrope cell organization within the pituitary gland.
Cell Death Discovery 9, (2023)
A balance of noncanonical Semaphorin signaling from the cerebrospinal fluid regulates apical cell dynamics during corticogenesis
Gerstmann K., Kindbeiter K., Telley L., et al.. 🔗 https://doi.org/10.1126/sciadv.abo4552
Résumé :
During corticogenesis, dynamic regulation of apical adhesion is fundamental to generate correct numbers and cell identities. While radial glial cells (RGCs) maintain basal and apical anchors, basal progenitors and neurons detach and settle at distal positions from the apical border. Whether diffusible signals delivered from the cerebrospinal fluid (CSF) contribute to the regulation of apical adhesion dynamics remains fully unknown. Secreted class 3 Semaphorins (Semas) trigger cell responses via Plexin-Neuropilin (Nrp) membrane receptor complexes. Here, we report that unconventional Sema3-Nrp preformed complexes are delivered by the CSF from sources including the choroid plexus to Plexin-expressing RGCs via their apical endfeet. Through analysis of mutant mouse models and various ex vivo assays mimicking ventricular delivery to RGCs, we found that two different complexes, Sema3B/Nrp2 and Sema3F/Nrp1, exert dual effects on apical endfeet dynamics, nuclei positioning, and RGC progeny. This reveals unexpected balance of CSF-delivered guidance molecules during cortical development.
Science Advances 8, (2022)
Environmental cues from neural crest derivatives act as metastatic triggers in an embryonic neuroblastoma model
Ben Amar D., Thoinet K., Villalard B., et al.. 🔗 https://doi.org/10.1038/s41467-022-30237-3
Résumé :
AbstractEmbryonic malignant transformation is concomitant to organogenesis, often affecting multipotent and migratory progenitors. While lineage relationships between malignant cells and their physiological counterparts are extensively investigated, the contribution of exogenous embryonic signals is not fully known. Neuroblastoma (NB) is a childhood malignancy of the peripheral nervous system arising from the embryonic trunk neural crest (NC) and characterized by heterogeneous and interconvertible tumor cell identities. Here, using experimental models mimicking the embryonic context coupled to proteomic and transcriptomic analyses, we show that signals released by embryonic sympathetic ganglia, including Olfactomedin-1, induce NB cells to shift from a noradrenergic to mesenchymal identity, and to activate a gene program promoting NB metastatic onset and dissemination. From this gene program, we extract a core signature specifically shared by metastatic cancers with NC origin. This reveals non-cell autonomous embryonic contributions regulating the plasticity of NB identities and setting pro-dissemination gene programs common to NC-derived cancers.
Nature Communications 13, (2022)
X-linked partial corpus callosum agenesis with mild intellectual disability: identification of a novel L1CAM pathogenic variant
Bousquet I., Bozon M., Castellani V., et al.. 🔗 https://doi.org/10.1007/s10048-020-00629-y
Résumé :
Résumé non disponible.
neurogenetics 22, 43-51 (2021)
SlitC-PlexinA1 mediates iterative inhibition for orderly passage of spinal commissural axons through the floor plate
Ducuing H., Gardette T., Pignata A., et al.. 🔗 https://doi.org/10.7554/elife.63205
Résumé :
Spinal commissural axon navigation across the midline in the floor plate requires repulsive forces from local Slit repellents. The long-held view is that Slits push growth cones forward and prevent them from turning back once they became sensitized to these cues after midline crossing. We analyzed with fluorescent reporters Slits distribution and FP glia morphology. We observed clusters of Slit-N and Slit-C fragments decorating a complex architecture of glial basal process ramifications. We found that PC2 proprotein convertase activity contributes to this pattern of ligands. Next, we studied Slit-C acting via PlexinA1 receptor shared with another FP repellent, the Semaphorin3B, through generation of a mouse model baring PlexinA1Y1815Fmutation abrogating SlitC but not Sema3B responsiveness, manipulations in the chicken embryo, and ex vivo live imaging. This revealed a guidance mechanism by which SlitC constantly limits growth cone exploration, imposing ordered and forward-directed progression through aligned corridors formed by FP basal ramifications.
eLife 9, (2020)
A Spatiotemporal Sequence of Sensitization to Slits and Semaphorins Orchestrates Commissural Axon Navigation
Pignata A., Ducuing H., Boubakar L., et al.. 🔗 https://doi.org/10.1016/j.celrep.2019.08.098
Résumé :
Résumé non disponible.
Cell Reports 29, 347-362.e5 (2019)
Hijacking of Embryonic Programs by Neural Crest-Derived Neuroblastoma: From Physiological Migration to Metastatic Dissemination
Delloye-Bourgeois C., Castellani V.. 🔗 https://doi.org/10.3389/fnmol.2019.00052
Résumé :
Résumé non disponible.
Frontiers in Molecular Neuroscience 12, (2019)
Molecular Memory of Morphologies by Septins during Neuron Generation Allows Early Polarity Inheritance
Boubakar L., Falk J., Ducuing H., et al.. 🔗 https://doi.org/10.1016/j.neuron.2017.07.027
Résumé :
Résumé non disponible.
Neuron 95, 834-851.e5 (2017)
Septin functions during neuro-development, a yeast perspective
Falk J., Boubakar L., Castellani V.. 🔗 https://doi.org/10.1016/j.conb.2019.01.012
Résumé :
Résumé non disponible.
Current Opinion in Neurobiology 57, 102-109 (2019)
Microenvironment-Driven Shift of Cohesion/Detachment Balance within Tumors Induces a Switch toward Metastasis in Neuroblastoma
Delloye-Bourgeois C., Bertin L., Thoinet K., et al.. 🔗 https://doi.org/10.1016/j.ccell.2017.09.006
Résumé :
Résumé non disponible.
Cancer Cell 32, 427-443.e8 (2017)
Genetic specification of left–right asymmetry in the diaphragm muscles and their motor innervation
Charoy C., Dinvaut S., Chaix Y., et al.. 🔗 https://doi.org/10.7554/elife.18481
Résumé :
The diaphragm muscle is essential for breathing in mammals. Its asymmetric elevation during contraction correlates with morphological features suggestive of inherent left–right (L/R) asymmetry. Whether this asymmetry is due to L versus R differences in the muscle or in the phrenic nerve activity is unknown. Here, we have combined the analysis of genetically modified mouse models with transcriptomic analysis to show that both the diaphragm muscle and phrenic nerves have asymmetries, which can be established independently of each other during early embryogenesis in pathway instructed by Nodal, a morphogen that also conveys asymmetry in other organs. We further found that phrenic motoneurons receive an early L/R genetic imprint, with L versus R differences both in Slit/Robo signaling and MMP2 activity and in the contribution of both pathways to establish phrenic nerve asymmetry. Our study therefore demonstrates L–R imprinting of spinal motoneurons and describes how L/R modulation of axon guidance signaling helps to match neural circuit formation to organ asymmetry.
eLife 6, (2017)
Cerebrospinal fluid-derived Semaphorin3B orients neuroepithelial cell divisions in the apicobasal axis
Arbeille E., Reynaud F., Sanyas I., et al.. 🔗 https://doi.org/10.1038/ncomms7366
Résumé :
Résumé non disponible.
Nature Communications 6, (2015)
PlexinA1 is a new Slit receptor and mediates axon guidance function of Slit C-terminal fragments
Delloye-Bourgeois C., Jacquier A., Charoy C., et al.. 🔗 https://doi.org/10.1038/nn.3893
Résumé :
Résumé non disponible.
Nature Neuroscience 18, 36-45 (2014)
Financements et soutiens



Financements passés