NeuraCel : Neurobiologie et vieillissement de C. elegans
Chef d’équipe : Jean-Louis BESSEREAU
C. elegans | synapse | jonction neuromusculaire | récepteurs de l’acétylcholine | vieillissement | génétique | neurobiologie | électrophysiologie optogénétique | microscopie photonique super résolutive | microscopie électronique
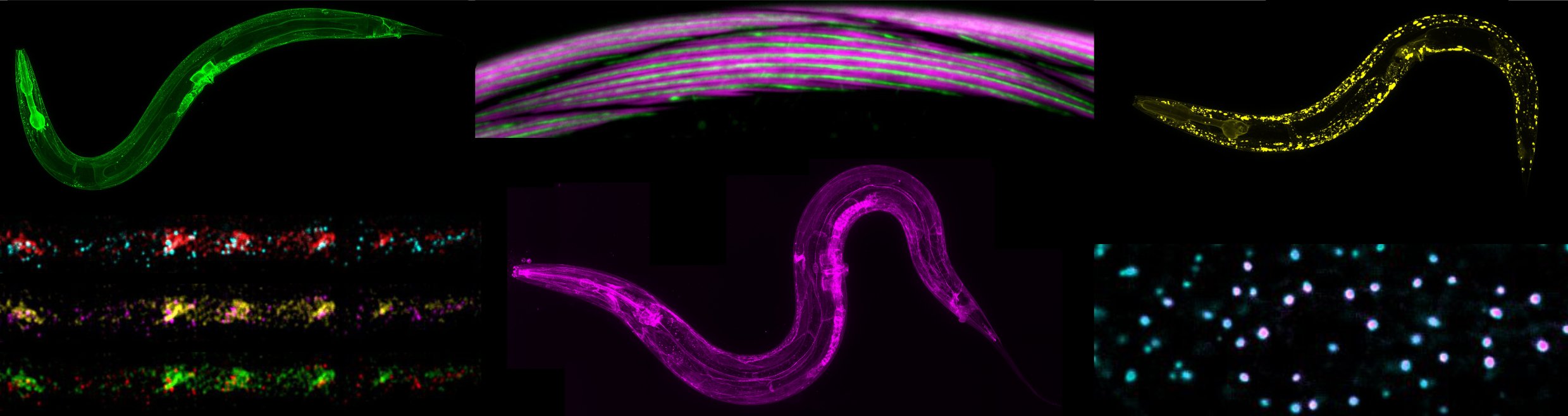
 Les synapses neurochimiques sont des nanomachines sophistiquées qui assurent le transfert et le traitement de l’information entre les neurones.
Les synapses neurochimiques sont des nanomachines sophistiquées qui assurent le transfert et le traitement de l’information entre les neurones.
Comme chaque neurone reçoit des milliers d’entrées synaptiques utilisant des neurotransmetteurs différents, sa membrane constitue une mosaïque de microdomaines spécialisés formés par des agrégats de récepteurs positionnés en face des sites de libération du neurotransmetteur. Notre laboratoire étudie les mécanismes cellulaires et moléculaires de la mise en place et du maintien des synapses, avec un intérêt particulier pour les processus qui contrôlent la biogenèse et la localisation des récepteurs des neurotransmetteurs en situations normale et pathologique.
Notre stratégie repose sur la combinaison d’approches de génétique, d’imagerie, d’électrophysiologie et de biochimie utilisant le ver rond Caenorhabditis elegans comme organisme d’expérimentation (pour plus d’information sur ce nématode, voir “Introduction aux Caenorhabditis elegans“). En utilisant la synapse neuromusculaire comme synapse modèle, nous avons identifié plusieurs gènes impliqués dans le positionnement des récepteurs de l’acétylcholine et du GABA. Parmi eux, nous avons mis en évidence un organisateur synaptique antérograde nécessaire à l’assemblage d’échafaudages protéiques extracellulaires piégeant les récepteurs. Nous analysons actuellement l’organisation, la dynamique et la maintenance de ces échafaudages, ainsi que les gènes impliqués dans la biosynthèse et l’adressage de ces récepteurs.
Nos résultats contribuent à une meilleure compréhension de la biologie de la synapse, en contextes physiologique et pathologique. En effet, depuis quelques années, il a été montré dans un nombre croissant de maladies neuropsychiatriques telles que l’autisme, la schizophrénie ou l’épilepsie, qu’il existe des dysfonctionnements synaptiques, faisant émerger le concept de « synaptopathies ». Par ailleurs, la fonction de nombreux gènes identifiés comme des facteurs de susceptibilité de maladies neuropsychiatriques reste inconnue, et il est probable que le paysage mutationnel de ces maladies se complexifie encore avec le foisonnement des données générées par l’accélération du séquençage des génomes de milliers de patients. Nous pensons que la simplicité d’un organisme tel que C. elegans est un outil précieux pour contribuer à démêler cette complexité.
Financements en cours
Financement ANR JCJC
SECRETS
Décodage de l’organisation synaptique : Comprendre le trafic et les voies de sécrétion des protéines d’échafaudage extracellulaires
2025-10-01

Financement ANR
BEAMS
Exploration des biomarqueurs, des modèles expérimentaux et de la signature immunitaire pour la maladie anti-IgLON5
2025-10-01

Financement ANR
AdheSyn
Organisation fonctionnelle et structurale des complexes d’adhésion synaptiques IgLONs
2025-10-01


Financement FRM Maladies Neurodégénératives
AIMING
Mieux comprendre les mécanismes de la neurodégénérescence dans la maladie anti-IgLON5
2025-01-01

Financement de la Fondation Maladies Rares
2024-04-24


Financement CEFIPRA
Analyse du trafic intracellulaire et de la sécrétion des organisateurs synaptiques extracellulaires
2023

Financement FRM
Analysis of cholinergic transmission using C. elegans genetics
2023-2026

Financement ANR
RESTOICH
Ionotropic receptor stoichiometry: pathways and pathology
2022-07-28


Financement AFM
Genetic control of acetylcholine receptor expression: from new mechanisms to functional genomics
2022-08-31
Our project on genetic control of AChR expression has been selected by AFM. We will try to get a comprehensive genetic landscape of We will perform new-generation screens for decreased amount of AChR at the C. elegans neuromuscular junction based on direct visualization of the receptors in vivo. Then we will test the functional conservation of C. elegans genes for AChR biosynthesis in mammals using advanced genome manipulation in mammalian cell lines. Finally we have developed a pipeline to go back from human gene variants to C. elegans and we will assess the pathogenicity of human polymorphisms found in genes required for AChR biosynthesis. modulators and shed new light on the cell biology of AChR.
Financement ANR
MUSAGE
Genetic and molecular determinants of Muscle Ageing
2021-10-01

Soutien du LABEX CORTEX

Membres de l’équipe
- Kshanti AFRICANO-GOMEZ — IE
- Jean-Louis BESSEREAU — PUPH, UCBL, HDR
- Lina BOUMASMOUD — IE
- Clara CHAPEL — Doctorante
- Mélissa CIZERON — CRCN, INSERM
- Manuela D’ALESSANDRO — CV — CRCN, INSERM
- Anaïs DUMAS — Doctorante
- Manon FROMENT — Doctorante
- Evan GOUY — Chercheur, HCL
- Laure GRANGER — AI, CNRS
- Maëlle JOSPIN — MCU, UCBL, HDR
- Driss LAABID — Technicien, UCBL
- Delphine LE GUERN — IE
- Suzanne LE REUN — Doctorante
- Greta MAIELLANO — Doctorante
- Laurent MOLIN — IE, CNRS
- Liubov PATRASH — Doctorante
- Bérangère PINAN-LUCARRÉ — CV — CRHC, INSERM, HDR
- Florence SOLARI — CRHC, INSERM, HDR
- Charlotte THIRE-CHEVREUSE — M2
- Alexandre VAUDANO — Doctorant
Publications récentes
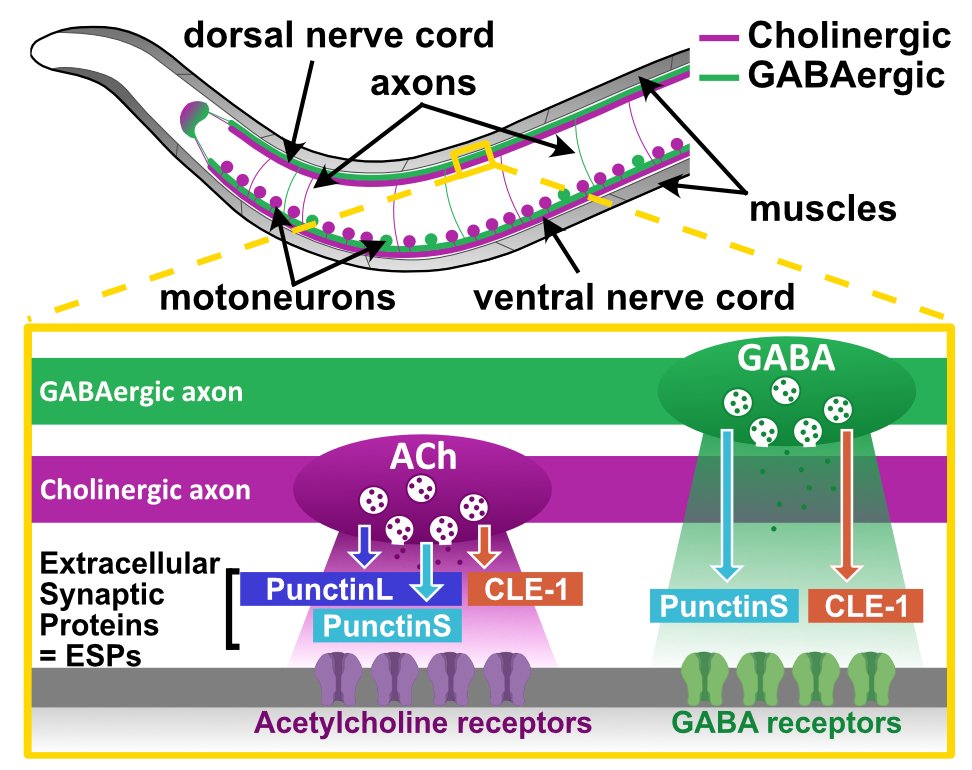
The nonfibrillar multiplexin collagen CLE-1 defines cholinergic synapse identity
Cizeron M., Dumas A., Le Reun S., et al.. 🔗 https://doi.org/10.1126/sciadv.adz1291
Résumé :
Fast neurotransmission requires the coordinated localization of neurotransmitter receptors opposite presynaptic release sites, which usually relies on the transsynaptic interaction of synaptic adhesion molecules. However, some extracellular proteins have been shown to coordinate pre- and postsynaptic differentiation by more elusive mechanisms. Here, we identify the nonfibrillar collagen CLE-1, a member of the evolutionarily conserved multiplexin family, as a master determinant of synapse identity in Caenorhabditis elegans . C. elegans muscle cells are innervated by both cholinergic and GABAergic motoneurons. The CLE-1B isoform is secreted by motoneurons and localizes to neuromuscular junctions. Loss of CLE-1B causes the relocalization of acetylcholine receptors to GABAergic synapses. We show that CLE-1B positions previously unidentified proteolytic fragments of the extracellular scaffold Punctin/MADD-4 to align acetylcholine receptors with cholinergic terminals and independently modulates receptor abundance. These findings reveal CLE-1 as a dual-function synaptic organizer that integrates spatial and quantitative control of postsynaptic receptor localization to maintain synapse identity.
Science Advances 11, (2025)
Biallelic CRELD1 variants cause severe muscle weakness and infantile epilepsy
D'Alessandro M., Bamborschke D., Bülow M., et al.. 🔗 https://doi.org/10.1093/braincomms/fcaf326
Résumé :
Abstract Nicotinic acetylcholine receptors are widely expressed in the peripheral and central nervous systems. Mutations in acetylcholine receptor-subunit genes have been associated with neuromuscular diseases, such as arthrogryposis multiplex congenita (AMC) and epilepsy. We report a patient with arthrogryposis, severe muscle weakness and neurodevelopmental delay. During his first year of life, he developed therapy-refractory epilepsy. Using whole-exome sequencing, we identified the compound pathogenic variants c. 875G>A (p. Cys292Tyr) and c. 959delA (p. Gln320Argfs*25) in the cysteine-rich with epidermal growth factor-like domain protein 1 gene (CRELD1, NM_001077415.3). Recently, functional studies have shown that CRELD1 is a membrane-associated endoplasmic reticulum-resident protein disulphide isomerase that acts as a maturation enhancer of AChR biogenesis, thereby controlling the abundance of functional receptors at the cell surface. To test pathogenicity, we took advantage of the genetics and extremely rapid genome editing in Caenorhabditis elegans. We were able to model these heterozygous variants and observed a decrease in AChRs at the neuromuscular junction. Hence, our study identifies compound heterozygous CRELD1 variants responsible for a rare neurodevelopmental disorder characterized by arthrogryposis, muscle weakness and epilepsy.
Brain Communications 7, (2025)
Axon termination of the SAB motor neurons in C. elegans depends on pre- and postsynaptic activity
Weinreb A., Nguyen M., Lemaitre Q., et al.. 🔗 https://doi.org/10.1101/2025.09.05.674448
Résumé :
Abstract Axon termination is a critical step in neural circuit formation, but the contribution of activity from postsynaptic targets to this process remains unclear. Using Caenorhabditis elegans SAB neurons as a model system, we showed that inhibition of muscle activity during a critical period of postembryonic development led to axonal overgrowth and ectopic synapse formation. This effect is mediated by a local retrograde signal and requires neuronal voltage-gated calcium channels (VGCCs) acting cell-autonomously to constrain axon growth. Manipulating SAB neuron excitability demonstrated that increased intrinsic neuronal activity drives overgrowth, while reducing activity suppresses it, establishing a functional link between muscle-derived cues and presynaptic excitability. Transcriptomic analysis and genetic studies further implicate the neuropeptides FLP-18 and NLP-12 as essential modulators of this activity-dependent process. Our findings reveal a temporally and spatially restricted retrograde signaling mechanism in motor neurons, where target activity, neuronal calcium dynamics and neuropeptide signaling cooperate to ensure proper axon termination. These results highlight conserved principles of activity-dependent regulation at neuromuscular junctions and provide a framework for understanding how motor circuits integrate target feedback to sculpt precise connectivity.
Journal non disponible , (2025)
A trans-synaptic IgLON adhesion molecular complex directly contacts and clusters a nicotinic receptor
Mialon M., Patrash L., Weinreb A., et al.. 🔗 https://doi.org/10.1101/2024.09.05.611427
Résumé :
SUMMARYThe localization and clustering of neurotransmitter receptors at appropriate postsynaptic sites is a key step in the control of synaptic transmission. Here, we identify a novel paradigm for the synaptic localization of an ionotropic acetylcholine receptor (AChR) based on the direct interaction of its extracellular domain with a cell adhesion molecule of the IgLON family. Our results show that RIG-5 and ZIG-8, which encode the sole IgLONs inC. elegans,are tethered in the pre- and postsynaptic membranes, respectively, and interactin vivothrough their first immunoglobulin-like (Ig) domains. In addition, ZIG-8 traps ACR-16 via a directcis-interaction between the ZIG-8 Ig2 domain and the base of the large extracellular AChR domain. Such mechanism has never been reported, but all these molecules are conserved during evolution. Similar interactions may directly couple Ig superfamily adhesion molecules and members of the large family of Cys-loop ionotropic receptors, including AChRs, in the mammalian nervous system, and may be relevant in the context of IgLON-associated brain diseases.
Journal non disponible , (2024)
UNC-30/PITX coordinates neurotransmitter identity with postsynaptic GABA receptor clustering
Correa E., Mialon M., Cizeron M., et al.. 🔗 https://doi.org/10.1242/dev.202733
Résumé :
ABSTRACT Terminal selectors are transcription factors that control neuronal identity by regulating expression of key effector molecules, such as neurotransmitter biosynthesis proteins and ion channels. Whether and how terminal selectors control neuronal connectivity is poorly understood. Here, we report that UNC-30 (PITX2/3), the terminal selector of GABA nerve cord motor neurons in Caenorhabditis elegans, is required for neurotransmitter receptor clustering, a hallmark of postsynaptic differentiation. Animals lacking unc-30 or madd-4B, the short isoform of the motor neuron-secreted synapse organizer madd-4 (punctin/ADAMTSL), display severe GABA receptor type A (GABAAR) clustering defects in postsynaptic muscle cells. Mechanistically, UNC-30 acts directly to induce and maintain transcription of madd-4B and GABA biosynthesis genes (e.g. unc-25/GAD, unc-47/VGAT). Hence, UNC-30 controls GABAA receptor clustering in postsynaptic muscle cells and GABA biosynthesis in presynaptic cells, transcriptionally coordinating two crucial processes for GABA neurotransmission. Further, we uncover multiple target genes and a dual role for UNC-30 as both an activator and a repressor of gene transcription. Our findings on UNC-30 function may contribute to our molecular understanding of human conditions, such as Axenfeld–Rieger syndrome, caused by PITX2 and PITX3 gene variants.
Development 151, (2024)
Array tomography of in vivo labeled synaptic receptors
Britz S., Luccardini C., Markert S., et al.. 🔗 https://doi.org/10.1016/bs.mcb.2024.02.029
Résumé :
Résumé non disponible.
Methods in Cell Biology , 139-174 (2024)
Calcineurin-Dependent Homeostatic Response of C. elegans Muscle Cells upon Prolonged Activation of Acetylcholine Receptors
Florin F., Bonneau B., Briseño-Roa L., et al.. 🔗 https://doi.org/10.3390/cells12172201
Résumé :
Pharmacological adaptation is a common phenomenon observed during prolonged drug exposure and often leads to drug resistance. Understanding the cellular events involved in adaptation could provide new strategies to circumvent this resistance issue. We used the nematode Caenorhabditis elegans to analyze the adaptation to levamisole, an ionotropic acetylcholine receptor agonist, used for decades to treat nematode parasitic infections. Genetic screens in C. elegans identified “adapting mutants” that initially paralyze upon exposure to levamisole as the wild type (WT), but recover locomotion after a few hours whereas WT remain paralyzed. Here, we show that levamisole induces a sustained increase in cytosolic calcium concentration in the muscle cells of adapting mutants, lasting several hours and preceding a decrease in levamisole-sensitive acetylcholine receptors (L-AChR) at the muscle plasma membrane. This decrease correlated with a drop in calcium concentration, a relaxation of the animal’s body and a resumption of locomotion. The decrease in calcium and L-AChR content depends on calcineurin activation in muscle cells. We also showed that levamisole adaptation triggers homeostatic mechanisms in muscle cells including mitochondria remodeling, lysosomal tubulation and an increase in autophagic activity. Levamisole adaptation thus provides a new experimental paradigm for studying how cells cope with calcium stress.
Cells 12, 2201 (2023)
Adamtsl3 mediates DCC signaling to selectively promote GABAergic synapse function
Cramer T., Pinan-Lucarre B., Cavaccini A., et al.. 🔗 https://doi.org/10.1016/j.celrep.2023.112947
Résumé :
Résumé non disponible.
Cell Reports 42, 112947 (2023)
Synaptogenesis: unmasking molecular mechanisms using Caenorhabditis elegans
Mizumoto K., Jin Y., Bessereau J.. 🔗 https://doi.org/10.1093/genetics/iyac176
Résumé :
Abstract The nematode Caenorhabditis elegans is a research model organism particularly suited to the mechanistic understanding of synapse genesis in the nervous system. Armed with powerful genetics, knowledge of complete connectomics, and modern genomics, studies using C. elegans have unveiled multiple key regulators in the formation of a functional synapse. Importantly, many signaling networks display remarkable conservation throughout animals, underscoring the contributions of C. elegans research to advance the understanding of our brain. In this chapter, we will review up-to-date information of the contribution of C. elegans to the understanding of chemical synapses, from structure to molecules and to synaptic remodeling.
GENETICS 223, (2023)
An extracellular scaffolding complex confers unusual rectification upon an ionotropic acetylcholine receptor in
C. elegans
Jospin M., Bonneau B., Lainé V., et al.. 🔗 https://doi.org/10.1073/pnas.2113545119
Résumé :
Biophysical properties of ligand-gated receptors can be profoundly modified by auxiliary subunits or by the lipid microenvironment of the membrane. Hence, it is sometimes challenging to relate the properties of receptors reconstituted in heterologous expression systems to those of their native counterparts. Here we show that the properties of Caenorhabditis elegans levamisole-sensitive acetylcholine receptors (L-AChRs), the ionotropic acetylcholine receptors targeted by the cholinergic anthelmintic levamisole at neuromuscular junctions, can be profoundly modified by their clustering machinery. We uncovered that L-AChRs exhibit a strong outward rectification in vivo, which was not previously described in heterologous systems. This unusual feature for an ionotropic AChR is abolished by disrupting the interaction of the receptors with the extracellular complex required for their synaptic clustering. When recorded at −60 mV, levamisole-induced currents are similar in the wild type and in L-AChR-clustering–defective mutants, while they are halved in these mutants at more depolarized physiological membrane potentials. Consequently, levamisole causes a strong muscle depolarization in the wild type, which leads to complete inactivation of the voltage-gated calcium channels and to an irreversible flaccid paralysis. In mutants defective for L-AChR clustering, the levamisole-induced depolarization is weaker, allowing voltage-gated calcium channels to remain partially active, which eventually leads to adaptation and survival of the worms. This explains why historical screens for C. elegans mutants resistant to levamisole identified the components of the L-AChR clustering machinery, in addition to proteins required for receptor biosynthesis or efficacy. This work further emphasizes the importance of pursuing ligand-gated channel characterization in their native environment.
Proceedings of the National Academy of Sciences 119, (2022)
DAF‐2/insulin IGF‐1 receptor regulates motility during aging by integrating opposite signaling from muscle and neuronal tissues
Roy C., Molin L., Alcolei A., et al.. 🔗 https://doi.org/10.1111/acel.13660
Résumé :
AbstractDuring aging, preservation of locomotion is generally considered an indicator of sustained good health, in elderlies and in animal models. In Caenorhabditis elegans, mutants of the insulin‐IGF‐1 receptor DAF2/IIRc represent a paradigm of healthy aging, as their increased lifespan is accompanied by a delay in age‐related loss of motility. Here, we investigated the DAF‐2/IIRc‐dependent relationship between longevity and motility using an auxin‐inducible degron to trigger tissue‐specific degradation of endogenous DAF‐2/IIRc. As previously reported, inactivation of DAF‐2/IIRc in neurons or intestine was sufficient to extend the lifespan of worms, whereas depletion in epidermis, germline, or muscle was not. However, neither intestinal nor neuronal depletion of DAF‐2/IIRc prevented the age‐related loss of motility. In 1‐day‐old adults, DAF‐2/IIRc depletion in neurons reduced motility in a DAF‐16/FOXO dependent manner, while muscle depletion had no effect. By contrast, DAF‐2 depletion in the muscle of middle‐age animals improved their motility independently of DAF‐16/FOXO but required UNC‐120/SRF. Yet, neuronal or muscle DAF‐2/IIRc depletion both preserved the mitochondria network in aging muscle. Overall, these results show that the motility pattern of daf‐2 mutants is determined by the sequential and opposing impact of neurons and muscle tissues and can be dissociated from the regulation of the lifespan. This work also provides the characterization of a versatile tool to analyze the tissue‐specific contribution of insulin‐like signaling in integrated phenotypes at the whole organism level.
Aging Cell 21, (2022)
Synapse Formation and Function Across Species: Ancient Roles for CCP, CUB, and TSP-1 Structural Domains
González-Calvo I., Cizeron M., Bessereau J., et al.. 🔗 https://doi.org/10.3389/fnins.2022.866444
Résumé :
The appearance of synapses was a crucial step in the creation of the variety of nervous systems that are found in the animal kingdom. With increased complexity of the organisms came a greater number of synaptic proteins. In this review we describe synaptic proteins that contain the structural domains CUB, CCP, or TSP-1. These domains are found in invertebrates and vertebrates, and CUB and CCP domains were initially described in proteins belonging to the complement system of innate immunity. Interestingly, they are found in synapses of the nematode C. elegans, which does not have a complement system, suggesting an ancient function. Comparison of the roles of CUB-, CCP-, and TSP-1 containing synaptic proteins in various species shows that in more complex nervous systems, these structural domains are combined with other domains and that there is partial conservation of their function. These three domains are thus basic building blocks of the synaptic architecture. Further studies of structural domains characteristic of synaptic proteins in invertebrates such as C. elegans and comparison of their role in mammals will help identify other conserved synaptic molecular building blocks. Furthermore, this type of functional comparison across species will also identify structural domains added during evolution in correlation with increased complexity, shedding light on mechanisms underlying cognition and brain diseases.
Frontiers in Neuroscience 16, (2022)
Evidence of a dual mechanism of action underlying the anti-proliferative and cytotoxic effects of ammonium-alkyloxy-stilbene-based α7- and α9-nicotinic ligands on glioblastoma cells
Pucci S., Bolchi C., Bavo F., et al.. 🔗 https://doi.org/10.1016/j.phrs.2021.105959
Résumé :
Résumé non disponible.
Pharmacological Research 175, 105959 (2022)
The HSPG syndecan is a core organizer of cholinergic synapses
Zhou X., Vachon C., Cizeron M., et al.. 🔗 https://doi.org/10.1083/jcb.202011144
Résumé :
The extracellular matrix has emerged as an active component of chemical synapses regulating synaptic formation, maintenance, and homeostasis. The heparan sulfate proteoglycan (HSPG) syndecans are known to regulate cellular and axonal migration in the brain. They are also enriched at synapses, but their synaptic functions remain more elusive. Here, we show that SDN-1, the sole orthologue of syndecan in C. elegans, is absolutely required for the synaptic clustering of homomeric α7-like acetylcholine receptors (AChRs) and regulates the synaptic content of heteromeric AChRs. SDN-1 is concentrated at neuromuscular junctions (NMJs) by the neurally secreted synaptic organizer Ce-Punctin/MADD-4, which also activates the transmembrane netrin receptor DCC. Those cooperatively recruit the FARP and CASK orthologues that localize α7-like-AChRs at cholinergic NMJs through physical interactions. Therefore, SDN-1 stands at the core of the cholinergic synapse organization by bridging the extracellular synaptic determinants to the intracellular synaptic scaffold that controls the postsynaptic receptor content.
Journal of Cell Biology 220, (2021)
Specific heparan sulfate modifications stabilize the synaptic organizer MADD-4/Punctin at Caenorhabditis elegans neuromuscular junctions
Cizeron M., Granger L., Bülow H., et al.. 🔗 https://doi.org/10.1093/genetics/iyab073
Résumé :
Abstract Heparan sulfate (HS) proteoglycans contribute to the structural organization of various neurochemical synapses. Depending on the system, their role involves either the core protein or the glycosaminoglycan chains. These linear sugar chains are extensively modified by HS modification enzymes, resulting in highly diverse molecules. Specific modifications of glycosaminoglycan chains may thus contribute to a sugar code involved in synapse specificity. Caenorhabditis elegans is particularly useful to address this question because of the low level of genomic redundancy of these enzymes, as opposed to mammals. Here, we systematically mutated the genes encoding HS modification enzymes in C. elegans and analyzed their impact on excitatory and inhibitory neuromuscular junctions (NMJs). Using single chain antibodies that recognize different HS modification patterns, we show in vivo that these two HS epitopes are carried by the SDN-1 core protein, the unique C. elegans syndecan ortholog, at NMJs. Intriguingly, these antibodies differentially bind to excitatory and inhibitory synapses, implying unique HS modification patterns at different NMJs. Moreover, while most enzymes are individually dispensable for proper organization of NMJs, we show that 3-O-sulfation of SDN-1 is required to maintain wild-type levels of the extracellular matrix protein MADD-4/Punctin, a central synaptic organizer that defines the identity of excitatory and inhibitory synaptic domains at the plasma membrane of muscle cells.
Genetics 218, (2021)
Sushi domain-containing protein 4 controls synaptic plasticity and motor learning
González-Calvo I., Iyer K., Carquin M., et al.. 🔗 https://doi.org/10.7554/elife.65712
Résumé :
Fine control of protein stoichiometry at synapses underlies brain function and plasticity. How proteostasis is controlled independently for each type of synaptic protein in a synapse-specific and activity-dependent manner remains unclear. Here, we show that Susd4 , a gene coding for a complement-related transmembrane protein, is expressed by many neuronal populations starting at the time of synapse formation. Constitutive loss-of-function of Susd4 in the mouse impairs motor coordination adaptation and learning, prevents long-term depression at cerebellar synapses, and leads to misregulation of activity-dependent AMPA receptor subunit GluA2 degradation. We identified several proteins with known roles in the regulation of AMPA receptor turnover, in particular ubiquitin ligases of the NEDD4 subfamily, as SUSD4 binding partners. Our findings shed light on the potential role of SUSD4 mutations in neurodevelopmental diseases.
eLife 10, (2021)
The Ig-like domain of Punctin/MADD-4 is the primary determinant for interaction with the ectodomain of neuroligin NLG-1
Platsaki S., Zhou X., Pinan-Lucarré B., et al.. 🔗 https://doi.org/10.1074/jbc.ra120.014591
Résumé :
Résumé non disponible.
Journal of Biological Chemistry 295, 16267-16279 (2020)
The netrin receptor UNC-40/DCC assembles a postsynaptic scaffold and sets the synaptic content of GABAA receptors
Zhou X., Gueydan M., Jospin M., et al.. 🔗 https://doi.org/10.1038/s41467-020-16473-5
Résumé :
AbstractIncreasing evidence indicates that guidance molecules used during development for cellular and axonal navigation also play roles in synapse maturation and homeostasis. In C. elegans the netrin receptor UNC-40/DCC controls the growth of dendritic-like muscle cell extensions towards motoneurons and is required to recruit type A GABA receptors (GABAARs) at inhibitory neuromuscular junctions. Here we show that activation of UNC-40 assembles an intracellular synaptic scaffold by physically interacting with FRM-3, a FERM protein orthologous to FARP1/2. FRM-3 then recruits LIN-2, the ortholog of CASK, that binds the synaptic adhesion molecule NLG-1/Neuroligin and physically connects GABAARs to prepositioned NLG-1 clusters. These processes are orchestrated by the synaptic organizer CePunctin/MADD-4, which controls the localization of GABAARs by positioning NLG-1/neuroligin at synapses and regulates the synaptic content of GABAARs through the UNC-40-dependent intracellular scaffold. Since DCC is detected at GABA synapses in mammals, DCC might also tune inhibitory neurotransmission in the mammalian brain.
Nature Communications 11, (2020)
Molecular Architecture of Genetically-Tractable GABA Synapses in C. elegans
Zhou X., Bessereau J.. 🔗 https://doi.org/10.3389/fnmol.2019.00304
Résumé :
Résumé non disponible.
Frontiers in Molecular Neuroscience 12, (2019)
CRELD1 is an evolutionarily-conserved maturational enhancer of ionotropic acetylcholine receptors
D'Alessandro M., Richard M., Stigloher C., et al.. 🔗 https://doi.org/10.7554/elife.39649
Résumé :
The assembly of neurotransmitter receptors in the endoplasmic reticulum limits the number of receptors delivered to the plasma membrane, ultimately controlling neurotransmitter sensitivity and synaptic transfer function. In a forward genetic screen conducted in the nematode C. elegans, we identified crld-1 as a gene required for the synaptic expression of ionotropic acetylcholine receptors (AChR). We demonstrated that the CRLD-1A isoform is a membrane-associated ER-resident protein disulfide isomerase (PDI). It physically interacts with AChRs and promotes the assembly of AChR subunits in the ER. Mutations of Creld1, the human ortholog of crld-1a, are responsible for developmental cardiac defects. We showed that Creld1 knockdown in mouse muscle cells decreased surface expression of AChRs and that expression of mouse Creld1 in C. elegans rescued crld-1a mutant phenotypes. Altogether these results identify a novel and evolutionarily-conserved maturational enhancer of AChR biogenesis, which controls the abundance of functional receptors at the cell surface.
eLife 7, (2018)
Mergoud dit Lamarche A., Molin L., Pierson L., et al.. 🔗 https://doi.org/10.1111/acel.12713
Résumé :
SummaryAging is commonly defined as the loss of global homeostasis, which results from progressive alteration of all organs function. This model is currently challenged by recent data showing that interventions that extend lifespan do not always increase the overall fitness of the organism. These data suggest the existence of tissue‐specific factors that regulate the pace of aging in a cell‐autonomous manner. Here, we investigated aging of Caenorhabditis elegans striated muscles at the subcellular and the physiological level. Our data show that muscle aging is characterized by a dramatic decrease in the expression of genes encoding proteins required for muscle contraction, followed by a change in mitochondria morphology, and an increase in autophagosome number. Myofilaments, however, remain unaffected during aging. We demonstrated that the conserved transcription factor UNC‐120/SRF regulates muscle aging biomarkers. Interestingly, the role of UNC‐120/SRF in the control of muscle aging can be dissociated from its broader effect on lifespan. In daf‐2/insulin/IGF1 receptor mutants, which exhibit a delayed appearance of muscle aging biomarkers and are long‐lived, disruption of unc‐120 accelerates muscle aging but does not suppress the lifespan phenotype of daf‐2 mutant. Conversely, unc‐120 overexpression delays muscle aging but does not increase lifespan. Overall, we demonstrate that UNC‐120/SRF controls the pace of muscle aging in a cell‐autonomous manner downstream of the insulin/IGF1 receptor.
Aging Cell 17, (2018)
Biallelic mutation of UNC50, encoding a protein involved in AChR trafficking, is responsible for arthrogryposis
Abiusi E., D’Alessandro M., Dieterich K., et al.. 🔗 https://doi.org/10.1093/hmg/ddx288
Résumé :
Résumé non disponible.
Human Molecular Genetics 26, 3989-3994 (2017)
Sélection de publications
C. elegans Punctin Clusters GABAA Receptors via Neuroligin Binding and UNC-40/DCC Recruitment
Tu H., Pinan-Lucarré B., Ji T., et al.. 🔗 https://doi.org/10.1016/j.neuron.2015.05.013
Résumé :
Résumé non disponible.
Neuron 86, 1407-1419 (2015)
C. elegans Punctin specifies cholinergic versus GABAergic identity of postsynaptic domains
Pinan-Lucarré B., Tu H., Pierron M., et al.. 🔗 https://doi.org/10.1038/nature13313
Résumé :
Résumé non disponible.
Nature 511, 466-470 (2014)
Biosynthesis of ionotropic acetylcholine receptors requires the evolutionarily conserved ER membrane complex
Richard M., Boulin T., Robert V., et al.. 🔗 https://doi.org/10.1073/pnas.1216154110
Résumé :
The number of nicotinic acetylcholine receptors (AChRs) present in the plasma membrane of muscle and neuronal cells is limited by the assembly of individual subunits into mature pentameric receptors. This process is usually inefficient, and a large number of the synthesized subunits are degraded by endoplasmic reticulum (ER)-associated degradation. To identify cellular factors required for the synthesis of AChRs, we performed a genetic screen in the nematode Caenorhabditis elegans for mutants with decreased sensitivity to the cholinergic agonist levamisole. We isolated a partial loss-of-function allele of ER membrane protein complex-6 (emc-6) , a previously uncharacterized gene in C. elegans . emc-6 encodes an evolutionarily conserved 111-aa protein with two predicted transmembrane domains. EMC-6 is ubiquitously expressed and localizes to the ER. Partial inhibition of EMC-6 caused decreased expression of heteromeric levamisole-sensitive AChRs by destabilizing unassembled subunits in the ER. Inhibition of emc-6 also reduced the expression of homomeric nicotine-sensitive AChRs and GABA A receptors in C. elegans muscle cells. emc-6 is orthologous to the yeast and human EMC6 genes that code for a component of the recently identified ER membrane complex (EMC). Our data suggest this complex is required for protein folding and is connected to ER-associated degradation. We demonstrated that inactivation of additional EMC members in C. elegans also impaired AChR synthesis and induced the unfolded protein response. These results suggest that the EMC is a component of the ER folding machinery. AChRs might provide a valuable proxy to decipher the function of the EMC further.
Proceedings of the National Academy of Sciences 110, (2013)
Positive modulation of a Cys-loop acetylcholine receptor by an auxiliary transmembrane subunit
Boulin T., Rapti G., Briseño-Roa L., et al.. 🔗 https://doi.org/10.1038/nn.3197
Résumé :
Résumé non disponible.
Nature Neuroscience 15, 1374-1381 (2012)
A secreted complement-control-related protein ensures acetylcholine receptor clustering
Gendrel M., Rapti G., Richmond J., et al.. 🔗 https://doi.org/10.1038/nature08430
Résumé :
Résumé non disponible.
Nature 461, 992-996 (2009)
Eight genes are required for functional reconstitution of the
Caenorhabditis elegans
levamisole-sensitive acetylcholine receptor
Boulin T., Gielen M., Richmond J., et al.. 🔗 https://doi.org/10.1073/pnas.0806933105
Résumé :
Levamisole-sensitive acetylcholine receptors (L-AChRs) are ligand-gated ion channels that mediate excitatory neurotransmission at the neuromuscular junctions of nematodes. They constitute a major drug target for anthelminthic treatments because they can be activated by nematode-specific cholinergic agonists such as levamisole. Genetic screens conducted in Caenorhabditis elegans for resistance to levamisole toxicity identified genes that are indispensable for the biosynthesis of L-AChRs. These include 5 genes encoding distinct AChR subunits and 3 genes coding for ancillary proteins involved in assembly and trafficking of the receptors. Despite extensive analysis of L-AChRs in vivo, pharmacological and biophysical characterization of these receptors has been greatly hampered by the absence of a heterologous expression system. Using Xenopus laevis oocytes, we were able to reconstitute functional L-AChRs by coexpressing the 5 distinct receptor subunits and the 3 ancillary proteins. Strikingly, this system recapitulates the genetic requirements for receptor expression in vivo because omission of any of these 8 genes dramatically impairs L-AChR expression. We demonstrate that 3 α- and 2 non-α-subunits assemble into the same receptor. Pharmacological analysis reveals that the prototypical cholinergic agonist nicotine is unable to activate L-AChRs but rather acts as a potent allosteric inhibitor. These results emphasize the role of ancillary proteins for efficient expression of recombinant neurotransmitter receptors and open the way for in vitro screening of novel anthelminthic agents.
Proceedings of the National Academy of Sciences 105, 18590-18595 (2008)
A transmembrane protein required for acetylcholine receptor clustering in Caenorhabditis elegans
Gally C., Eimer S., Richmond J., et al.. 🔗 https://doi.org/10.1038/nature02893
Résumé :
Résumé non disponible.
Nature 431, 578-582 (2004)






