SPARK – Signaling Pathways And Regulators of K+ channels
Principal investigator: Thomas Boulin — Team website
C. elegans | cellular excitability | potassium channels | polarity | rare disease | molecular & cellular neurobiology | CRISPR/Cas9 genome engineering
Research Topics and Main Projects
Polarity and Membrane Architecture of C. elegans Muscle Cells
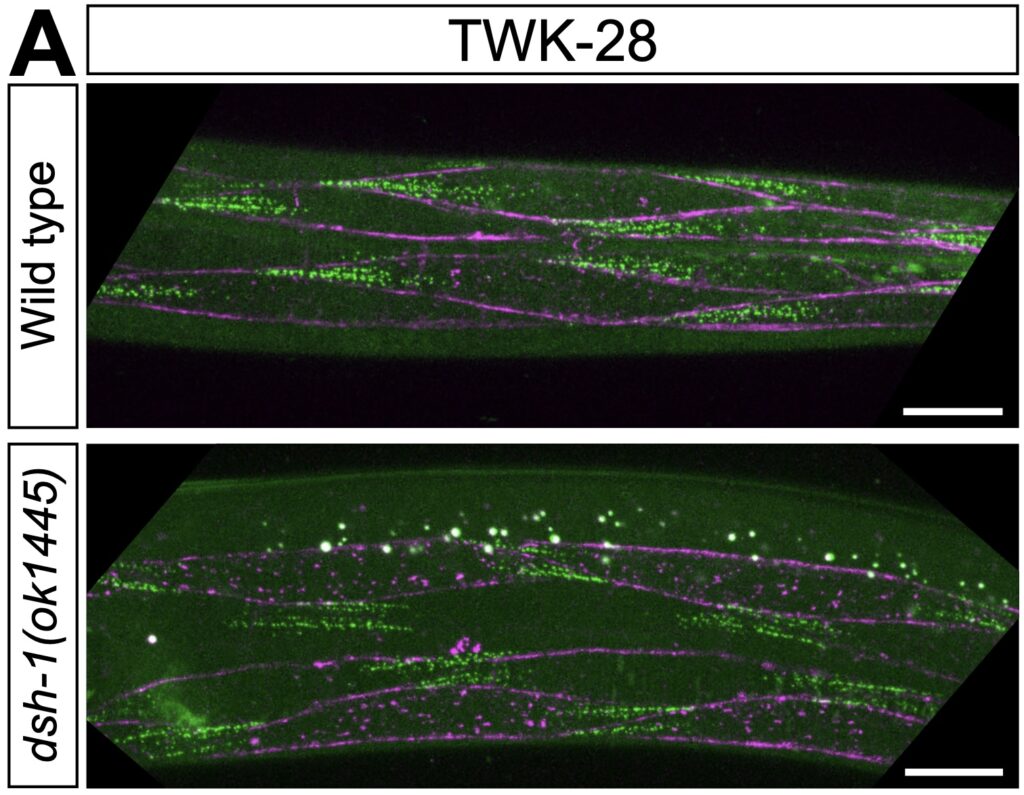
Cell polarity mechanisms create specialized membrane domains with unique protein compositions and functions. For example, most ion channels are addressed to specific compartments within excitable cells. By systematically analysing the localization of potassium channels and other membrane proteins, we recently discovered that the plasma membrane of C. elegans muscle cells is polarized (Peysson et al., Nature Communications, 2024).
Despite extensive study of C. elegans muscle development, this striking form of cellular polarization had gone unnoticed. It relies not on classical planar polarity proteins but on a non-canonical Wnt–Ror–Dvl pathway, a conserved and poorly understood signaling module also implicated in morphogenesis and cancer. In addition, we also discovered that the dystrophin-associated protein complex (DAPC) organizes distinct membrane sub-compartments within the sarcolemma.
Key questions we are addressing now are : (1) What are the downstream effectors of the Wnt-Ror-Dvl signaling pathway that implement muscle polarity? (2) How are distinct membrane domains established and how are their specific protein compositions determined?
Functional Analysis of Human Genetic Variants of K+ Channels and Regulatory Proteins Implicated in Rare Neurodevelopmental and Cardiovascular Diseases


Rare human diseases are only rare in name: more than 300 million people are affected by approximately 7,000 such conditions. For most patients, a years-long diagnostic odyssey is still the norm. With the rise of genomic sequencing, clinicians are now identifying an ever-increasing number of genetic variants. While frequently observed variants can often be interpreted easily, rare cases and de novo missense mutations remain particularly challenging. Functional validation studies have therefore become essential for determining the pathogenicity of variants found in patients with rare genetic disorders.
We use C. elegans as a robust model system to functionally validate variants of unknown significance (VUS) in potassium channels and their regulatory proteins, which are associated with rare neurodevelopmental disorders and hereditary cardiac arrhythmias (Boulin et al., MGM 2021; Delinière et al., Journal of Physiology, 2025). By employing CRISPR/Cas9 genome editing and phenotypic assays, we can assess the biological impact of VUS in a native cellular context.
Conducted in collaboration with clinicians at Columbia University and the Centre National de Référence des Troubles du Rythme Cardiaque d’Origine Héréditaire de Lyon (CERA, HCL), these projects connect our basic scientific research with clinically relevant applications. By combining clinical investigations with molecular, cellular, and model organism studies, we aim to resolve diagnostic uncertainties and uncover the mechanisms underlying rare human diseases.
Congenital long QT syndrome (LQTS) is a hereditary condition leading to sudden death in young individuals. With a prevalence of about 1/2000 births, it is the most common inherited channelopathy. Sixteen genes are linked to LQTS, and variants in the potassium channel gene KCNH2 alone account for 30–45% of cases. KCNH2 encodes the hERG subunit of the Kv11.1 channel, which carries the main repolarizing current in cardiomyocytes. hERG dysfunction prolongs repolarization and can trigger fatal ventricular arrhythmias.
We have recently described the clinical and electrophysiological features of the novel p.Gly603Ser KCNH2 variant (Delinière et al., Gene 2024). This patient mutation is the first homozygous loss-of-function variant in the Kv11.1 pore to cause a severe yet viable long-QT syndrome with delayed clinical expression.
Why Individuals Differ: Genetic, Developmental, and Environmental Drivers

Why individuals are different is a fundamental question in biology. Indeed, both genetic and non-genetic factors generate phenotypic variation. We address this basic question in C. elegans by combining computational analyses with experimental systems biology. We especially focus on genome-wide gene expression dynamics – including at single-cell and single-individual level – as a multidimensional information-rich intermediate phenotype and as a powerful generator of mechanistic hypotheses. To this end, we develop computational methods to infer and precisely quantify interpretable phenotypes from -omics data, such as physiological age (Bulteau and Francesconi, Nature Methods, 2022).
Using these strategies, we investigate how gene expression is regulated in space (tissues) and time (development and aging). We also study why genetically identical individuals differ, focusing in particular on how developmental history and maternal age shape phenotypic variation. Finally, we study how sensory perception of the environment in one generation can alter development, behavior, and fitness in the next, uncovering mechanisms of information transmission across generations.
Shaping the Electrical Identity of C. elegans Neurons – ETN Nervspan

Throughout development and aging, neurons undergo a series of morphological and functional changes to establish and maintain their identity and function. Recent advances in single-cell transcriptomics have revealed that distinct neuronal classes within the C. elegans nervous system express unique combinations of potassium channels. These specific ion channel profiles determine the biophysical and electrical properties that define neuron-specific functions throughout the organism’s lifespan.
We aim to understand (1) how distinct neuronal classes establish their unique potassium channel expression profiles, (2) the mechanisms that maintain this electrical identity over time, and (3) whether it can be influenced by external perturbations or shaped by sex-specific differences.
SPARK News
New publication in The Journal of Physiology – Caenorhabditis elegans as an in vivo model system for human inherited primary arrhythmia syndromes
2025-12-05

New publication in Brain Communications – Functional validation of human SK channels variants causing NEDMAB and Zimmermann–Laband syndrome-3 in C. elegans
2025-09-16

Congratulations to Sara for her first PhD publication!
Small conductance Ca2+-activated K+ channels (SK channels) are widely expressed in the central nervous system, where they play a crucial role in modulating neuronal excitability. Recent studies have identified missense variants in the genes encoding SK2 and SK3 channels as the cause of two rare neurodevelopmental disorders: NEDMAB and ZLS3, respectively.
Here, we used Caenorhabditis elegans as an in vivo model to investigate the functional consequences of these patient variants. The C. elegans orthologue KCNL-1 regulates neuronal and muscle excitability in the egg-laying system, a well-characterized model circuit. To visualize KCNL-1 expression and localization, we generated a fluorescent translational reporter at the endogenous kcnl-1 locus. We then introduced eight point mutations corresponding to pathogenic variants reported in NEDMAB or ZLS3 patients. Our study confirmed the molecular pathogenicity of the ZLS3-associated mutations, revealing a gain-of-function effect that led to increased in utero egg retention, likely due to electrical silencing of the egg-laying circuitry. NEDMAB mutations exhibited more complex phenotypic effects. Most caused a loss-of-function phenotype, indistinguishable from null mutants, while one displayed a clear gain-of-function effect. Additionally, a subset of NEDMAB variants altered KCNL-1 localization, suggesting an impairment in channel biosynthesis, trafficking or stability.
These findings provide new insights into the molecular mechanisms underlying NEDMAB and ZLS3 physiopathology, enhancing our understanding of SK channel dysfunction in human disease. Moreover, they establish C. elegans as a robust and cost-effective in vivo model for rapid functional validation of new SK channel mutations, paving the way for future investigations.
Congratulations, Olga, on being elected a junior member of the Institut Universitaire de France!
2025-05-15

New publication in PNAS – Constitutive sodium permeability in a C. elegans two-pore domain potassium channel.
2024-10-15

Congratulations to all involved in this multi-disciplinary exploration of the molecular determinants of K2P channel selectivity.
Potassium channels play a central role in modulating cellular excitability, particularly of neuronal cells. Their unique structure determines their ability to let ions pass selectively through cell membranes. The impact of pathological or evolutionary variations in this selectivity filter remains difficult to predict. Here, we reveal that UNC-58, a member of the two-pore domain potassium (K2P) channel family of C. elegans, exhibits an unusual sodium permeability due to a unique cysteine residue in its selectivity filter. Our findings underscore the importance of functional studies to determine how sequence variation in potassium channel selectivity filters can shape the electrical profiles of excitable cells.
New publication – Wnt-Ror-Dvl signalling and the dystrophin complex organize planar-polarized membrane compartments in C. elegans muscles
2024-06-10

Our study revealing the remarkably complex organisation of the worm’s sarcolemma is now out at Nature Communications!
Almost six years in the making, congratulations to Alice, Nora, Marie, Amandine, Noémie, Elise and Olga for this magnum opus revealing this entirely unsuspected new case of planar cell polarity in C. elegans muscles.
SUMMARY
Cell polarity mechanisms allow the formation of specialized membrane domains with unique protein compositions, signalling properties, and functional characteristics. By analyzing the localization of potassium channels and proteins belonging to the dystrophin-associated protein complex, we reveal the existence of distinct planar-polarized membrane compartments at the surface of C. elegans muscle cells. We find that muscle polarity is controlled by a non-canonical Wnt signalling cascade involving the ligand EGL-20/Wnt, the receptor CAM-1/Ror, and the intracellular effector DSH-1/Dishevelled. Interestingly, classical planar cell polarity proteins are not required for this process. Using time-resolved protein degradation, we demonstrate that –while it is essentially in place by the end of embryogenesis– muscle polarity is a dynamic state, requiring continued presence of DSH-1 throughout post-embryonic life. Our results reveal the unsuspected complexity of the C. elegans muscle membrane and establish a genetically tractable model system to study cellular polarity and membrane compartmentalization in vivo.
New publication – Functional and clinical characterization of a novel homozygous KCNH2 missense variant in the pore region of Kv11.1 leading to a viable but severe long-QT syndrome
2023-12-21
Congratulations to Antoine, Olga and the team of Philippe Chevalier (Rhythmology department, HCL Lyon) for our first collaborative study about Kv11.1/hERG potassium channels.
Highlights
- Homozygous missense variants in the pore of Kv11.1 often lead to intrauterine death.
- We describe a novel hERG p.Gly603Ser homozygous loss-of-function variant.
- It causes a severe but viable long-QT syndrome with a delayed clinical expression.
- Family segregation and functional analysis classify hERG p.Gly603Ser as probably pathogenic.
Ongoing Research Grants
DYSCO – Dystrophin-associated protein complex and subcellular compartmentalization
2023-07-13
Our project with the team of Vincent Mirouse (iGred, Clermont-Ferrand) and Helge Amthor (UVSQ) has been selected by ANR ! The Dystrophin Associated Protein Complex (DAPC) is a key actor of the cell – extracellular matrix (ECM) interface, as revealed by its implication in human genetic disorders. However, its molecular and cellular functions are still poorly understood because tractable model systems allowing state-of-the-art in vivo cell biology and genetic approaches are lacking. Our three teams have developed the first transgenic dystrophin reporters that have revealed remarkably compartmentalized membrane distributions of the DAPC in epithelia and muscle cells in C. elegans, Drosophila, and mouse. The project aims to elucidate the organization and dynamics of the DAPC and to characterize its new functions in relation to specific cell cortical compartments.
Fleggsibility – Dissecting the microevolutionary flexibility of a neural circuit
2022-08-13
Our project with the team of Christian Braendle (iBV Nice) and ViewPoint: Behavior Analysis Technologies has been selected by ANR ! On the heals of our joint publication (Vigne et al., Science Advances 2021) we will be trying to understand how specific neural circuits evolve to generate natural behavioural variation within species. In particular, the precise genetic changes that modulate cellular and developmental architectures of reproductive systems remain unclear. Here we will focus on the simple egg-laying circuit of the nematode Caenorhabditis elegans as a powerful model system to study natural microevolutionary (intraspecific) variability.
Join the team
Research Context

Questions that we would like to solve
- What are the cellular mechanisms that control the distribution of K+ channels in vivo in C. elegans?
- Are these pathways conserved in other organisms, including vertebrates?
- How are the number and the activity of K+ channels at the cell surface controlled?
- Are there specific factors that differentially regulate K+ channels in distinct tissues or cell types?
- Do regulators of K+ channels also control the biology of other ion channel families?
Functional Studies of C. elegans Two-Pore domain Potassium (K2P) Channels

Potassium channels are crucial for regulating cellular excitability, with their unique structure enabling selective ion passage through cell membranes. Gain‐of‐function mutations that modulate the activity of ion channels are essential tools to study their function. We have shown that a single conserved residue controls the activity of K2P potassium channels in vertebrates and invertebrates and can be mutated to progressively increase channel open probability (Ben Soussia et al., Nature Communications 2019). This discovery has been pivotal for our projects, as it enabled us to generate the necessary gain‐of‐function knockin lines to study the contribution of K2P channels to the control of cellular excitability.
Three studies stemming directly from this work have been published. Two collaborative studies describe the role of the TWK-40 channel in controlling locomotor and rhythmic behaviors (Yue et al., PNAS Nexus 2024; Meng et al., Science Advances 2024). In Meng et al., we describe a new conceptual framework for understanding how animals transition smoothly between opposing motor states, such as forward and backward movement. In the third study, we describe the functional characterization of an atypical potassium channel, UNC-58, which is the first example of a K2P channel that is constitutively permeable to sodium ions (Andrini et al., PNAS 2024).
Evolutionary Variation in Egg-Laying Behavior of C. elegans
Reproductive behaviors vary considerably among animals—not only between species but also within a single species. However, we still have a limited understanding of the molecular processes that allow neural circuits to evolve within a species to generate such behavioural variation.
We used the egg-laying circuit of C. elegans as a model system to study natural intraspecific microevolutionary variability. In collaboration with Christian Braendle’s team (iBV, Nice), we demonstrated that a mutation in a single gene, the SK potassium channel KCNL-1, results in the loss of environmental sensitivity in several natural isolates. Instead of regulating egg-laying based on the presence or absence of food, these isolates constitutively retain their embryos, which leads to internal hatching. By combining our complementary expertise, we achieved a comprehensive characterization—from the molecular and cellular levels to the evolutionary process – that led to the selection of this paradoxical phenotypic trait (Vigne et al., Science Advances 2021).
Building on this work, we published a second study that characterizes the intraspecific variation of egg-laying behavior in C. elegans (Mignerot et al., eLife 2023). In this study, by analysing a collection of approximately 300 wild isolates, we describe highly variable intra-uterine retention. By applying various pharmacological agents and genetically manipulating endogenous serotonin levels in 10 wild isolates, we demonstrated that this behavioural variation stems from an evolutionarily divergent neuromodulatory architecture.
Team Members
- Thomas BOULIN — CV — DR2 CNRS, HDR
- Olga ANDRINI CV — Associate professor, UCBL, HDR
- Antoine DELINIERE — Adjunct researcher, MD, PhD, Hôpital Cardiologique, HCL
- Mirko FRANCESCONI CV— CRCN, Inserm, HDR
- Kathrin GIESELER — PREX, UCBL, HDR
- Sandra DUPERRIER — AI, CNRS
- Laura AGRESTI — PhD candidate
- Parichitran AYYAMPERUMAL — PhD candidate
- Clara BLANCFUNEY — Post-doc
- Élise CHEYNET — PhD candidate
- Sara SECHI — PhD candidate
- Minh Thu NGUYEN — PhD candidate
Alumni
- Mélissa ZOUAK
- Claire LECROISEY
- Marie GENDREL
- Marine MERCIER
- Ismail BEN SOUSSIA
- Sonia EL MOURIDI
- Philippe TARDY
- Caroline BORKOWSKI
- Nora ZARIOHI
- Estèle LAFONT
- Alice LECLERCQ-BLONDEL
- Alice PEYSSON
- Guillaume BRULÈRE
Selected publications
Caenorhabditis elegans
as an
in vivo
model system for human inherited primary arrhythmia syndromes
Delinière A., Boulin T., Jospin M., et al.. 🔗 https://doi.org/10.1113/jp289661
Résumé :
Abstract Inherited primary arrhythmia syndromes (IPAS) are genetic heart diseases associated with an elevated risk of sudden cardiac death, particularly in young individuals. Modelling these rare and serious conditions is essential to elucidate their mechanisms and to identify new treatments. Most genes involved in IPAS (e.g., congenital long‐QT syndrome, catecholaminergic polymorphic ventricular tachycardia, calcium‐release deficiency syndrome, Andersen‐Tawil syndrome, Timothy syndrome, calmodulinopathies, and short‐QT syndrome) are conserved in Caenorhabditis elegans , a model organism that offers powerful genetic tools for precise gene manipulation, including knock‐in, knock‐out, and knock‐down approaches. In vivo studies in C. elegans can be used to characterize the consequences of genetic variants (at molecular, cellular, tissue, and behavioural scales), to identify new regulatory proteins, and to perform drug testing. Here we summarize the characteristics of human IPAS and highlight the accumulating evidence that supports the utility of C. elegans as a simple yet powerful in vivo model for these diseases, capable of filling the gap between in vitro studies and complex transgenic animal models. image
The Journal of Physiology , (2025)
Functional validation of human SK channels variants causing NEDMAB and Zimmermann–Laband syndrome-3 in C. elegans
Sechi S., Galaup C., Jospin M., et al.. 🔗 https://doi.org/10.1093/braincomms/fcaf351
Résumé :
Abstract Small conductance Ca2+-activated K+ channels (SK channels) are widely expressed in the central nervous system, where they play a crucial role in modulating neuronal excitability. Recent studies have identified missense variants in the genes encoding SK2 and SK3 channels as the cause of two rare neurodevelopmental disorders: NEDMAB and ZLS3, respectively. Here, we used Caenorhabditis elegans as an in vivo model to investigate the functional consequences of these patient variants. The C. elegans orthologue KCNL-1 regulates neuronal and muscle excitability in the egg-laying system, a well-characterized model circuit. To visualize KCNL-1 expression and localization, we generated a fluorescent translational reporter at the endogenous kcnl-1 locus. We then introduced eight point mutations corresponding to pathogenic variants reported in NEDMAB or ZLS3 patients. Our study confirmed the molecular pathogenicity of the ZLS3-associated mutations, revealing a gain-of-function effect that led to increased in utero egg retention, likely due to electrical silencing of the egg-laying circuitry. NEDMAB mutations exhibited more complex phenotypic effects. Most caused a loss-of-function phenotype, indistinguishable from null mutants, while one displayed a clear gain-of-function effect. Additionally, a subset of NEDMAB variants altered KCNL-1 localization, suggesting an impairment in channel biosynthesis, trafficking or stability. These findings provide new insights into the molecular mechanisms underlying NEDMAB and ZLS3 physiopathology, enhancing our understanding of SK channel dysfunction in human disease. Moreover, they establish C. elegans as a robust and cost-effective in vivo model for rapid functional validation of new SK channel mutations, paving the way for future investigations.
Brain Communications 7, (2025)
Constitutive sodium permeability in a
Caenorhabditis elegans
two-pore domain potassium channel
Andrini O., Ben Soussia I., Tardy P., et al.. 🔗 https://doi.org/10.1073/pnas.2400650121
Résumé :
Two-pore domain potassium (K2P) channels play a central role in modulating cellular excitability and neuronal function. The unique structure of the selectivity filter in K2P and other potassium channels determines their ability to allow the selective passage of potassium ions across cell membranes. The nematode C. elegans has one of the largest K2P families, with 47 subunit-coding genes. This remarkable expansion has been accompanied by the evolution of atypical selectivity filter sequences that diverge from the canonical TxGYG motif. Whether and how this sequence variation may impact the function of K2P channels has not been investigated so far. Here, we show that the UNC-58 K2P channel is constitutively permeable to sodium ions and that a cysteine residue in its selectivity filter is responsible for this atypical behavior. Indeed, by performing in vivo electrophysiological recordings and Ca 2+ imaging experiments, we demonstrate that UNC-58 has a depolarizing effect in muscles and sensory neurons. Consistently, unc-58 gain-of-function mutants are hypercontracted, unlike the relaxed phenotype observed in hyperactive mutants of many neuromuscular K2P channels. Finally, by combining molecular dynamics simulations with functional studies in Xenopus laevis oocytes, we show that the atypical cysteine residue plays a key role in the unconventional sodium permeability of UNC-58. As predicting the consequences of selectivity filter sequence variations in silico remains a major challenge, our study illustrates how functional experiments are essential to determine the contribution of such unusual potassium channels to the electrical profile of excitable cells.
Proceedings of the National Academy of Sciences 121, (2024)
Wnt-Ror-Dvl signalling and the dystrophin complex organize planar-polarized membrane compartments in C. elegans muscles
Peysson A., Zariohi N., Gendrel M., et al.. 🔗 https://doi.org/10.1038/s41467-024-49154-8
Résumé :
AbstractCell polarity mechanisms allow the formation of specialized membrane domains with unique protein compositions, signalling properties, and functional characteristics. By analyzing the localization of potassium channels and proteins belonging to the dystrophin-associated protein complex, we reveal the existence of distinct planar-polarized membrane compartments at the surface of C. elegans muscle cells. We find that muscle polarity is controlled by a non-canonical Wnt signalling cascade involving the ligand EGL-20/Wnt, the receptor CAM-1/Ror, and the intracellular effector DSH-1/Dishevelled. Interestingly, classical planar cell polarity proteins are not required for this process. Using time-resolved protein degradation, we demonstrate that –while it is essentially in place by the end of embryogenesis– muscle polarity is a dynamic state, requiring continued presence of DSH-1 throughout post-embryonic life. Our results reveal the unsuspected complexity of the C. elegans muscle membrane and establish a genetically tractable model system to study cellular polarity and membrane compartmentalization in vivo.
Nature Communications 15, (2024)
A tonically active master neuron modulates mutually exclusive motor states at two timescales
Meng J., Ahamed T., Yu B., et al.. 🔗 https://doi.org/10.1126/sciadv.adk0002
Résumé :
Continuity of behaviors requires animals to make smooth transitions between mutually exclusive behavioral states. Neural principles that govern these transitions are not well understood. Caenorhabditis elegans spontaneously switch between two opposite motor states, forward and backward movement, a phenomenon thought to reflect the reciprocal inhibition between interneurons AVB and AVA. Here, we report that spontaneous locomotion and their corresponding motor circuits are not separately controlled. AVA and AVB are neither functionally equivalent nor strictly reciprocally inhibitory. AVA, but not AVB, maintains a depolarized membrane potential. While AVA phasically inhibits the forward promoting interneuron AVB at a fast timescale, it maintains a tonic, extrasynaptic excitation on AVB over the longer timescale. We propose that AVA, with tonic and phasic activity of opposite polarities on different timescales, acts as a master neuron to break the symmetry between the underlying forward and backward motor circuits. This master neuron model offers a parsimonious solution for sustained locomotion consisted of mutually exclusive motor states.
Science Advances 10, (2024)
A leak K+ channel TWK-40 sustains the rhythmic motor program
Yue Z., Li Y., Yu B., et al.. 🔗 https://doi.org/10.1093/pnasnexus/pgae234
Résumé :
Abstract Leak potassium (K+) currents, conducted by two-pore domain K+ (K2P) channels, are critical for the stabilization of the membrane potential. The effect of K2P channels on motor rhythm remains enigmatic. We show here that the K2P TWK-40 contributes to the rhythmic defecation motor program (DMP) in Caenorhabditis elegans. Disrupting TWK-40 suppresses the expulsion defects of nlp-40 and aex-2 mutants. By contrast, a gain-of-function (gf) mutant of twk-40 significantly reduces the expulsion frequency per DMP cycle. In situ whole-cell patch clamping demonstrates that TWK-40 forms an outward current that hyperpolarize the resting membrane potential of dorsorectal ganglion ventral process B (DVB), an excitatory GABAergic motor neuron that activates expulsion muscle contraction. In addition, TWK-40 substantially contributes to the rhythmic activity of DVB. Specifically, DVB Ca2+ oscillations exhibit obvious defects in loss-of-function (lf) mutant of twk-40. Expression of TWK-40(gf) in DVB recapitulates the expulsion deficiency of the twk-40(gf) mutant, and inhibits DVB Ca2+ oscillations in both wild-type and twk-40(lf) animals. Moreover, DVB innervated enteric muscles also exhibit rhythmic Ca2+ defects in twk-40 mutants. In summary, these findings establish TWK-40 as a crucial neuronal stabilizer of DMP, linking leak K2P channels with rhythmic motor activity.
PNAS Nexus 3, (2024)
Functional analysis of a de novo variant in the neurodevelopment and generalized epilepsy disease gene NBEA
Boulin T., Itani O., El Mouridi S., et al.. 🔗 https://doi.org/10.1016/j.ymgme.2021.07.013
Résumé :
Résumé non disponible.
Molecular Genetics and Metabolism 134, 195-202 (2021)
A single-nucleotide change underlies the genetic assimilation of a plastic trait
Vigne P., Gimond C., Ferrari C., et al.. 🔗 https://doi.org/10.1126/sciadv.abd9941
Résumé :
A single-nucleotide change underlies the transition from environmentally induced to genetically encoded matricidal hatching.
Science Advances 7, (2021)
Mutation of a single residue promotes gating of vertebrate and invertebrate two-pore domain potassium channels
Ben Soussia I., El Mouridi S., Kang D., et al.. 🔗 https://doi.org/10.1038/s41467-019-08710-3
Résumé :
AbstractMutations that modulate the activity of ion channels are essential tools to understand the biophysical determinants that control their gating. Here, we reveal the conserved role played by a single amino acid position (TM2.6) located in the second transmembrane domain of two-pore domain potassium (K2P) channels. Mutations of TM2.6 to aspartate or asparagine increase channel activity for all vertebrate K2P channels. Using two-electrode voltage-clamp and single-channel recording techniques, we find that mutation of TM2.6 promotes channel gating via the selectivity filter gate and increases single channel open probability. Furthermore, channel gating can be progressively tuned by using different amino acid substitutions. Finally, we show that the role of TM2.6 was conserved during evolution by rationally designing gain-of-function mutations in four Caenorhabditis elegans K2P channels using CRISPR/Cas9 gene editing. This study thus describes a simple and powerful strategy to systematically manipulate the activity of an entire family of potassium channels.
Nature Communications 10, (2019)
Funding & Support
2023-2027 MSCA European Doctoral Network — « NERVSPAN – Molecular plasticity of neurons during C. elegans lifespan »
2023-2028 Agence Nationale de la Recherche — « DYSCO – Dystrophin-associated protein complex and subcellular compartmentalization »
2022-2026 Agence Nationale de la Recherche — « Fleggsibility – Dissecting the microevolutionary flexibility of a neural circuit »
2020-2025 Agence Nationale de la Recherche — « NBelegAns – Molecular, cellular, and clinical investigation of the autism, epilepsy, and neurodevelopmental disorder gene Neurobeachin/NBEA »
2020-2025 Labex CORTEX website
2016-2021 AFM Téléthon — Alliance MyoNeurALP
2019-2020 Fondation Maladies Rares — « Modeling disease-causing mutations of Neurobeachin/NBEA in the nematode Caenorhabditis elegans »
2013-2019 European Research Council — Kelegans — « Genetics and cell biology of K2P channels »
2013-2014 Fondation Fyssen













